Authors: M. Mac-Mary, J. Sainthillier, P. Creidi, J.P. Series, F. Vix, Ph. Humbert
- Journal: CARD (Annual Congress of Dermatological Research) meeting in Brest, France
- Date: 2005
Abstract
Introduction
GliSODin has already demonstrated activation properties on the principal internal antioxidants, in particular Superoxide Dismutase (SOD), Catalase and Glutathione Peroxidase. The purpose of this trial was to assess its efficacy in solar prevention and protection.
Materials and methods
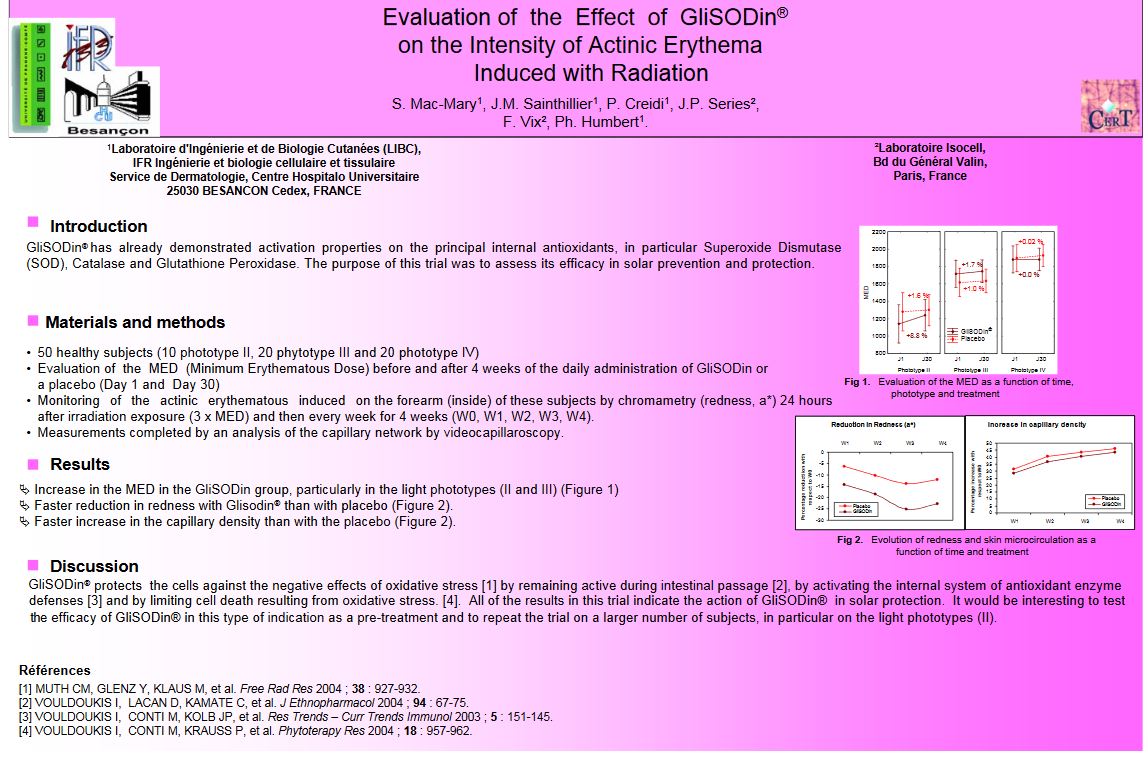
- 50 healthy subjects (10 phototype II, 20 phytotype III and 20 phototype IV)• Evaluation of the MED (Minimum Erythematous Dose)nbefore and after 4 weeks of the daily administration of GliSODin oral placebo (Day 1 and Day 30)
- Monitoring of the actinic erythematous induced on the forearm (inside)of these subjects by chromametry (redness, a*) 24 hoursafter irradiation exposure (3 x MED) and then every week for 4 weeks (W0, W1, W2, W3, W4).
- Measurements completed by an analysis of the capillarynetwork by videocapillaroscopy.
Results
- Increase in the MED in the GliSODin group, particularly in the light phototypes (II and III)
- Faster reduction in redness with Glisodin than with placebo.
- Faster increase in the capillary density than with the placebo.
Discussion
GliSODin protects the cells against the negative effects of oxidative stress by remaining active during intestinal passage, by activating the internal system of antioxidant enzyme defenses and by limiting cell death resulting from oxidative stress.
All of the results in this trial indicate the action of GliSODin in solar protection. It would be interesting to testthe efficacy of GliSODin in this type of indication as a pre-treatmentand to repeat the trial on a larger number of subjects, in particular on the light phototypes (II).
Categories: 7. Skin health & UV protection, Scientific studies
Share this post












